This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.

Laundry wastewater treatment in nuclear sector
Nucleantech was founded in 1992. Nowadays we have offices in Spain, France, England, USA and Mexico. During this time, more than 400 projects in 30 different countries have been carried out, 4 patents have been registered, and more than 320 treatment plants have been designed and built.
Nucleantech designs, supplies, and builds wastewater treatment plants using the best available technologies for wastewater minimization and reuse. Also, we reduce air emissions to a level that is both safe and compliant with legislation for release into the atmosphere.
Nucleantech provides turnkey solutions and takes a global approach to each and every project by means of the following issues:
Implementation of engineering solutions
Laboratory tests
Pilot plants for industrial tests
Turnkey facilities
Technical assistance and commissioning
2
Low-level radiation waste generated in nuclear sector
Nuclear waste is produced at source, during the uranium extraction, generated in the nuclear fuel plant and nuclear power plant operation when the nuclear fuel is used, and also some nuclear wastes are formed in radioisotope applications in medicine, industry, research, etc.
Nucleantech provides turnkey solutions and takes a global approach to each and every project by means of the following issues:
2.1
Uranium extraction
The uranium extraction can be done either in sulfuric media or in nitric media. Both procedures are used but the wastes generated are different.
Uranium extraction in sulfuric media
In the uranium extraction in sulfuric media, the crushed mineral is treated with diluted sulfuric acid to extract the uranium as UO2SO4. To oxidize uranium IV to VI, MnO2 or NaClO3 is used. The lixiviation process produces mining tailings as a waste. These mining tailings contain decay products from 238U, such as 234U (average life span of 250.000 years), 230Th (average life span of 75.320 years), 226Ra (average life span of 1.602 years), 222Rn (average life span of 3.8 days), 210Pb (average life span of 22 years). 80% of the 222Rn remains in tailing particles, but the other 20% is released into the atmosphere as it is a gas. The estimated activity in the Rn tailing wastes is around 300-1.000 pCi/m2·s.
The UO2SO4 solution has a concentration of around 0,3 and 0,8 g/L and it can be concentrated following two different ways
Nucleantech provides turnkey solutions and takes a global approach to each and every project by means of the following issues:
By using anionic resins. The species [UO2(SO4)2-] and [UO2(SO4)3-] are fixed to the anionic resin. They are eluted from the resins by means of a calcium chloride or a calcium nitrate solution.
Alkaline precipitation. Uranium concentrate is precipitated in alkaline media using ammonium hydroxide or magnesium hydroxide to obtain the Yellow Cake (uranate).
Waste is produced in the resins elution and in the alkaline media precipitate. This waste incorporates traces of 238U daughters.
Uranium extraction in nitric media
In the uranium extraction in nitric media, the mineral is treated with nitric acid to extract uranium as the dissolved salt UO2(NO3)2. In this process mining tailings are produced, which contain 238U daughters. The UO2(NO3)2 solution is mixed with TBP (tributyl phosphate), as the uranium compound is extracted with the organic phase. UO2(NO3)2 is re-extracted from the organic phase using pure water and UO2(NO3)3· 6 H2O is obtained (which is called UNH).
Therefore, in the uranium extraction the following nuclear wastes are produced:
226Ra(): it is a matter of concern due to its high dissolution facility in soils and, therefore, in groundwater.
230Th() and 222Ra(): the most problematic wastes regarding atmospheric inhalation. 222Ra() activity in the atmosphere is 300-1.000 pCi/m2s.
214Bi (): which is transformed into 210-Tl and 214Po
210Pb(): which is transformed into 210
During the whole extraction process, and activity control has to be carried out.
2.2
Uranium enrichment and nuclear fuel production
In this process, the fuel reactor (UO2) is prepared from the uranium concentrate obtained in the mining process (U3O8). The enrichment process consists of the following stages:
Yellow Cake or UNH dissolution in nitric acid solution to obtain a uranyl nitrate solution (UO2(NO3)2. Liquid-liquid extraction using TBP is carried out and subsequently is eluted with water to obtain pure (UO2(NO3)2.

Uranyl nitrate hexahydrate is obtained by means of evaporation. Then, the procedure has two possible ways to proceed: dry or wet methods
– Dry method: calcination of the uranyl nitrate to obtain UO3.
– Wet method: [U2O7(NH4)2] is obtained by precipitation and for thermal decomposition UO3 is formed.
Next, UO3 is reduced to UO2 using H2. Then, in a first step, hydrofluoriration using HF at 400 ºC is carried out, and in a second step, fluorination using F2 allows the obtainment of UF6.

The separation between isotopes 235U and 238U can be carried out either by separation cascade (gaseous diffusion) or by ultracentrifugation. Subsequently, both isotopes are mixed in the appropriate proportion (3,5% – 4,5% of 235U). To obtain nuclear fuel in the form of enriched UO2, UF6 is reduced using Mg0 or Ca0 to form uranium, which is then oxidized to UO2. 10 mm UO2 pellets are introduced in the zircaloid shells sealed and pressurized with helium gas.
Therefore, in the nuclear fuel production the following nuclear wastes are produced:
CaF2: which is formed in the treatment of HF and fluorine compounds with Ca(OH)2. Theses solid wastes contain uranium traces and its daughters. The amount of waste is around 26 Tm/1000 MWe·year and occupies a volume of 8.4 m3. The activity associated is about 0.6 Ci of uranium/1000 MWe·year.
Liquid wastes produced contain fluorides, uranium traces and uranium (235U and 238U) daughters. Its production is around 3200 m3/1000 MWe·year and its activity around 20 mCi of uranium/1000 MWe·year, 10 mCi of 234Th/1000 MWe·year, and 10 mCi of 234Pa/1000 MWe·year. These liquid wastes are produced in cleaning operations and in the UF6 cylinder washing process.
2.3
Nuclear power plant operation
In a nuclear power plant three different kind of waste are produced: operating waste (low and intermediate level), spent fuel waste (high level), and decommissioning waste (low and intermediate level). The management of high level waste will not be treated here.
Low and intermediate level waste (LILW) come from objects that are contaminated by contact with radioactive products but that are not radioactive in themselves, such as gloves used to handle them. Regulations state that the radioactivity of LILW must be below 10 mCi/kg (0.37 GBq). The majority of the radioactive atoms in solid LILW waste are beta emitters with a radioactive half-life of less than 30 years, such as 60Co. The radioactivity of these short-lived elements will have virtually disappeared in 300 years’ time because it will have decreased by a factor of at least 1000 over this period. Restricting this category to only short-lived radioelements means that it can include waste that is initially intermediate-level. Despite being referred to as short-lived, solid LILW does in fact contain some long-lived elements that would be too difficult or costly to remove completely. They are alpha emitters, which are toxic if ingested but are not very mobile. By law, the concentration of alpha emitters must not exceed one hundredth of the limit of 10 mCi/kg.
LILW comes from many different sources. For the most part, this waste is generated during maintenance operations (equipment, tools, cleaning rags, laundry wastewater, etc.) or during the operation of facilities, such as the waste from the treatment of liquid or gaseous effluents at nuclear facilities (filters and resins from the treatment of the reactor coolant liquid). It can also come from dismantling operations.
In the primary circuit coolant of a PWR type reactor the following chemical species can be distinguished:
Those which come from the fission fragments. This waste is formed in the fission reaction and it can be disseminated through the cracks of the fuel shells and arrive to the coolant liquid. Mainly, these species are 137Cs, 90Sr and 129I, among others. Waste which contains these species are high level waste.

Those which come from the zircaloy shell. Atoms of the zircaloy shell can be activated due to the neutron flux.

Uranium’s daughters. Uranium has the rare ability to shed protons and neutrons from its nucleus in the form of radioactive decay. The result of this decay creates an entirely different element. The elements in Uranium’s ‘family tree’ are referred to as ‘Daughters’. Uranium’s decay chain creates a total of 14 different elements, all radioactive save the last, lead which does not decay further. The most important is 239Pu for its long life (29400 years) and its high toxicity. Also, uranium’s daughters can be disseminated through the cracks of the fuel shells and arrive to the coolant liquid. Waste which contains these species are high level waste.

Activated products from structural elements. The primary cooling circuit has a huge heat exchange surface, around 10.000 m2, made of Inconel (75% Ni and 25% Cr, with 59Co impurities). These structural elements are activated with the neutron flux and produce isotopes of low and intermediate level, which leave the structure by corrosion. 14C arises from CO2 activation (from the air dissolved in the coolant) and 3H appears as a water proton activation.
3
Laundry wastewater generation and conventional treatment
In nuclear power plants laundry wastewater is produced in the protective clothing washing which has been used in the restricted areas. Protective clothing, gloves, footwear and protection masks are used as a barrier protection against particles, sprays or liquid spills which could contain radionuclides.
All of these materials are disposed in containers to be washed and then reused. The wastewater generated in the washing process, apart from the organic material and detergent, may contain radioactive particles and radioactive dissolved substances.
The radioactive liquid laundry waste is generated, not only in decontamination of equipment and washing of contaminated cloths, but also in staff showers.
The radioactivity of the laundry waste is ranged between 10-7-10-4 Ci/mL, but it’s total radioactivity cannot be disregarded because its yearly generation volume, around 4,000 ton/1,000 MWe, is significant. Only for South Korea, which has a net capacity of 22,505 MWe (source: World Nuclear Association, www.world-nuclear.org), the radioactive liquid laundry generation represents around 90,000 ton/year.
The conventional treatment of this waste is described below. The laundry effluent is discharged to a volume control tank, where its radioactivity is measured and pH is adjusted to precipitate metals that could be in dissolution. Also, coagulant and flocculant are added to precipitate suspended particles. Once the sludge is separated by filtration, the treated water is accumulated in a volume control tank to measure its radioactivity.
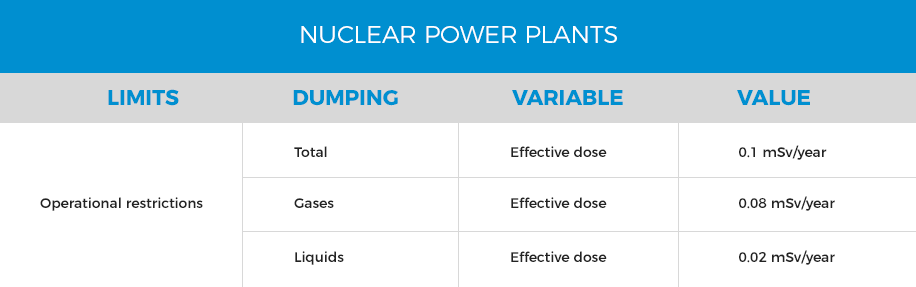
If the effluent does not present radioactivity or its radioactivity level complies with legal regulations 10CFR20, the effluent can be discharged to public waterways. The dumping limits for radioactive effluents are presented in the following table:
If the processed water contains radionuclides with activity higher than that allowed by the regulator, a treatment is required. Once the process water is filtered, and the precipitation sludge is separated, it is introduced into a forced convection evaporator.
In the evaporation process, values of activity concentration factor between 10 and 50 and decontamination factors around 105 are reached. In the evaporator a concentrate is obtained with the salts present in the solution and possible particles that have not previously precipitated, in addition to a condensed vapor that needs to be controlled. Finally, the controlled process water is dumped into public waterways if it complies with the regulations.
At this point, the formation of scale on the internal wall of the evaporation equipment, which could retain salts with activity, should be taken into account. The vapours are condensed in a volumetric tank that allows their radioactive activity to be determined. At the same time, gases that require a specific treatment are separated. In the steam generated in the condenser there is the possibility of generation of gaseous effluents transporting gases such as 3H, Xe, Kr and 14C in the form of CO2.
To remove the particles transported with the steam and certain gases (iodine), the gas mixture is circulated through activated carbon columns. The gases (3H, noble gases) that could be transported by the steam and have a short half-life are stored in a decay tank in order to reduce their activity and are finally discharged into the atmosphere, after regulator controlling. Discharges of liquid effluents with radioactive material must comply with the instantaneous limits of activity and the operational restriction of doses established in the nuclear facility, as well as the annual dose limits established by the relevant regulator. The total dose limit between liquid and gaseous effluent is 0.1mSv/year and an annual equivalent dose limit for the skin and any other body considered individually of 50 mSv in any period of 12 consecutive months.
4
Nucleantech’s solution for laundry wastewater treatment
The objective is the treatment of laundry waters from a nuclear installation taking into account the characteristics of this industrial effluent. From the treatment point of view, the requirement is lower in relation to other types of industrial laundry. The waters coming from a laundry of a nuclear facility in which protective clothing is washed have low values of COD and BOD5, coming from its use by the staff, values of conductivity due to salts, presence of grease and oils that come from maintenance work and low suspended particle values. However, at this point it has been taking into account that there is the possibility of incorporating radioactive particles from the installation into the washing waters (60Co, 90Sr, 137Cs, 129I, 131I, etc.) that have attached in protective clothing. In this sense, a possible treatment for this contamination by radioactive particles must be foreseen. On the other hand, the number of staff in a nuclear power plant is very variable depending on the stops, refuelling, maintenance, etc. The objective is to minimize water consumption by recycling it, obtaining a quality cleaning both from the textile and radiochemical point of view and generating the minimum volume of waste.
Some processes documented for the treatment of the radioactive liquid laundry waste use RO to avoid the problems resulting from the treatment by evaporation of the non-degradable detergents. However, the RO process produces a high rejection flow rate, which constitutes another waste that must be treated prior to its discharge.
The process developed by Nucleantech uses vacuum evaporation -as nuclear power plants have inexpensive sources of heat- to reduce to the minimum the waste generation and avoid the problem of the degradation of detergents by using an amount of detergents 3 times less than conventional processes, due to the use of ozone during the washing process.
4.1
NUCLEANTECH® LAUNDRY
The first step consists of collecting the material to be washed, classifying the different materials and evaluating the activity of each team. It is important to check that there are no high radioactive activity values in the wash. In these anomalous cases, the protective clothing must be treated as low and intermediate activity waste (LILW), by compaction.
The washing process begins with the classification of the material and the evaluation of its radioactive activity. Afterwards they are introduced into the washing machine and the washing is carried out at room temperature. During the washing cycle, a small amount of ozone is added with several purposes. First, ozone dilates fibres and helps the penetration of detergent. Second, it allows the concentration of detergent to be lower than in a conventional wash (4 g/L rather than 10-12 g/L). Third, washing with ozone allows it to be carried out at room temperature without excessively altering the pH, remaining in a neutral medium. In addition, the amount of water needed compared to a conventional wash is reduced. Finally, the use of ozone in the washing allows the reduction of other adjuvants and, consequently, decreases the load of contaminants in the laundry wastewater. As in the conventional washing process, in the wastewater, apart from a certain organic load, particles in suspension, grease and oil will all appear.
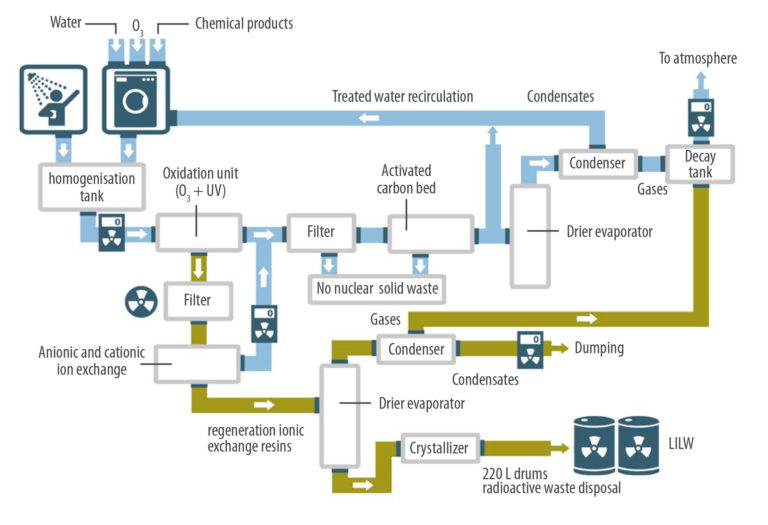
Once the washing is finished, the material is dried and the effluent is evacuated to a homogenization tank, where it is mixed with the staff shower wastewater.
The effluent from the homogenisation tank is subjected to an oxidation process using ozone and ultraviolet radiation. The objective of this stage is to decrease notably the organic load, disinfect and ionize possible radioactive metallic particles. Ozone, due to its oxidation capacity, eliminates organic matter (grease, sweat …) and odours. Ozone oxidation is carried out following two different ways. First, molecular ozone acts by direct oxidation of organic compounds. In a second way, the oxidation of organic matter can also be done by free hydroxyl radicals generated by ozone. Moreover, ozone has a disinfectant function since it acts on the wall of the microorganisms causing its destruction.
On the other hand, the working environment of a nuclear installation can cause certain operations where radioactive particles are fixed in protective clothing. Although this clothing contains an antistatic treatment, it cannot be discarded that in the washing water, activity is detected due to the presence of radioactive particles, such as 60Co, 54Mn and so on. These metallic particles, due to the effect of ozone, are oxidized to the state of ion, a form in which it is easier to reach their retention.
Depending on whether the effluent of the homogenization tank has high values of radioactivity (> 0.02 mSv / year) or not, the treatment will vary from this point. If radioactivity values higher than 0.02 mSv / year are not detected, the following stages follow the oxidation process:
Filtration
Filtration, with the goal of retaining the solids present in the wastewater and material that may have precipitated.
Adsorption
Adsorption with activated carbon, with the objective of reducing the COD, the adsorption of ozonides generated in the process and the capture of particles in suspension. In the case of the presence of radioactive particles in suspension, the activated carbon would act physically adsorbing them.
Vacuum evaporation
Vacuum evaporation, with the aim of transforming the effluent into a quality water stream that is reused in the laundry. The volatile outlet of the evaporator is cooled to condense the water and separate it from the gases present in the effluent. While the condensed water is recirculated to the laundry, the gases are released into the atmosphere before passing through a decay tank, to reduce the activity of possible radioactive gases. The concentrate of the evaporator that has not been evaporated is recirculated backwards so that it goes through the filtration process again.
Note that, if there is no radiation in the effluent of the homogenizer tank, all the waste produced (filter cake and washing of the active carbon column) can be managed following conventional procedures.
In the case that the effluent coming from the homogenization tank contains radioactivity higher than 0.02 mSv / year, the output of the oxidation stage is diverted to another treatment line, which is formed by the following stages:
- Filtration, to retain all possible solids present in the water and extend the life of the ion exchange columns of the next treatment stage.
- Ionic exchange through cationic and anionic columns. The objective is the retention in the exchange resins of the radioactive cations and anions in solution. The resins retain and remove them from the system, decreasing very significantly the radiation of the effluent at the exit of this stage. These retained ions are 60Co, 129I, 131I, 90Sr, 55Mn, 59Fe, 137Cs, 134Cs, 51Cr, etc.
- Vacuum evaporation, with the purpose of reducing the volume of the effluent generated in the regeneration process of the cationic and anion exchange columns. The evaporated effluent is cooled, the condensate stream can be dumped after checking the radioactivity level and the non-condensed gases are fed into the previously detailed decay tank. These gases can contain 222Rn, 3H2 and activated noble gases. The evaporator concentrate, which contains and concentrates all the radioactive species, undergoes a crystallization process to further reduce its volume. The dry solid that is extracted from the crystallizer must be managed following the approved procedures for the management of low and intermediate radiation waste (LILW) -confinement with concrete in 220L drums-.
4.2
Benefits in relation to conventional treatments
The treatment process of the laundry wastewater of a nuclear facility proposed by Nucleantech implies the introduction of exclusive advantages in relation to conventional processes. These advantages are summarized below:
High efficiency in the washing process
The use of ozone in the washing allows the washing to be carried out with a lower concentration of detergent, compared to conventional washes (4 g/L compared to the usual 12 g/L). In addition, its double mechanism of action, direct oxidation by molecular ozone and the generation of hydroxyl ions, produces an increase in efficiency and an important reduction of other chemical products used in the washing (neutralizers, softeners, optical brighteners, etc.). It acts effectively on sweat residue, odours of textiles, dilating fibres and helping the penetration of detergents. Ozone in water is eliminated by forming oxygen, so that the concentration of oxygen in solution increases, favouring the cleaning potential of the detergents used. Ozone biocidal action has been contrasted in laundries for groups such as hospitals and retirement homes. It is necessary to take into account the fluctuating staff of a nuclear power station (350 people during normal operating periods, in refuel times it can triple its number), this fact requires flexibility in the washing process. In this case, ozone allows this flexibility to be modulated by varying the concentration in the washing circuit.
The elimination of ozone, which does not generate any residue and forms oxygen, allows a high oxygen concentration in solution. This fact allows washing under room temperature conditions, without the need to use hot water. Therefore, it allows energy savings and savings in the wearing of protective clothing, taking into account that it has a polymeric base and the action of a high temperature can degrade its protection capacity.
The disinfection treatment is another key value of the use of ozone in the laundry process. With a redox potential of 2.07V, ozone exceeds chlorine in oxidation capacity. This effect causes the destruction of the cell wall of the microorganisms. The oxidation capacity is shown in the breakdown of the C-N bond, which causes the depolymerization in the nucleic chains of the bacteria. Ozone is effective in the elimination of bacteria, viruses, protozoa, nematodes, fungi, cell aggregates, and spores. On the other hand, it acts at the cytoplasm level, oxidizing internal mechanisms. This directly affects the reproduction of the bacterial flora.

Reuse of wash water
The wastewater, once it has been treated, has a quality such that it is possible to reuse it in the washing process. In addition to saving water in the process, the minimization of water consumption considerably reduces the amount of effluents discharged abroad, which, in a nuclear facility, is a parameter of key importance.

High reduction of waste production in the process
This treatment process is characterized by achieving a huge reduction in the volume of low and medium activity wastes generated that must be managed in accordance with regulated procedures, which have a high cost. This supposes a great economic saving in comparison with other processes.

Forecast and control of radioactive activity
The liquid laundry waste may contain radioactive particles from the working environment of a nuclear power plant (60Co, 129I, 131I, 90Sr, 55Mn, 59Fe, 137Cs, 134Cs, 51Cr, etc.). These particles are incorporated into the washing wastewater increasing its radioactive activity. In order to control this process, ozone oxidizes and ionizes these particles so that the mixed ion exchange resins (anionic and cationic) retain the ions in solution. In the event that the particles do not fully oxidize, they would be retained in the activated carbon filter. In this way, it is possible to keep the level of activity in the wastewater under control.

Flexibility of the treatment process to the presence of activity
The wastewater treatment process has been designed to adapt to the presence or absence of activity. The process consists of two lines of treatment, the basic one and the activity one. If there are no radioactive species in the water, only the basic treatment line is used, saving energy and resources. In the case of detecting radioactivity, the wastewater is diverted to the activity treatment line, and once the radioactivity has been eliminated, the water is returned to the basic treatment line.

Flexibility of the process to important fluctuations of flow and load
In nuclear power plants, depending on whether they are in the process of being stopped or started or if they are operating in a steady state, the number of workers present in the facilities can vary significantly. The variability in the size of the staff directly affects the amount of wastewater generated by the laundry and showers. In addition, when there are stops, the necessary maintenance work is carried out, so the pollutant load increases markedly. The treatment proposed by Nucleantech presents a high adaptation to variations both in the quantity and the composition of the wastewater produced.

Strict compliance with the most demanding regulations
All the technology used in the process is proven technology that ensures that, in addition to ensuring high efficiency in the washing process and minimizing the economic costs of treatment, effluents are discharged abroad fulfilling the requirements of the strictest regulations.
5
Other Nucleantech treatment processes for nuclear wastes
Nucleantech has developed other treatment processes addressed to the minimization of nuclear wastes, such as the treatment of the purges of the secondary cooling circuit in the PWR nuclear power plants, the treatment of the wastewater generated in the production of UF6 in the uranium enrichment process or the treatment and minimization of low radioactivity solid waste generated in the nuclear facilities by means of pyrolysis. These processes are described below.
5.1
NUCLEANTECH® H3BO3

Nucleantech technology provides an efficient solution for the treatment of the purges of the secondary cooling circuit in the PWR nuclear power plants.
Moreover, this solution allows the recovery of the boric acid dissolved in the primary cooling circuit in the PWR nuclear power plants.
This solution ensures maximum efficiency in the recovery of boric acid of nuclear quality. On the other hand, it helps to reduce the volume of liquid waste generated, with the consequent reduction in the cost of waste management.
5.2
NUCLEANTECH® UF6

This process has the goal of treating the wastewater generated in the production of UF6 in the uranium enrichment process.
This solution recovers the water used to wash the UF6 cylinders, while allowing the minimization of the solid waste produced.
This solid waste reduction is obtained by means of a high efficiency vacuum evaporator followed by a vacuum crystalliser.
This system reduces the volume of waste, which has an economic impact on the cost of managing this waste.
5.3
NUCLEANTECH® NWDR

This process provides a solution to the treatment and minimization of low radioactivity solid waste generated in nuclear facilities, such as gloves, masks, glasses, work clothes, caps, wood of pallets, etc., by means of pyrolysis.
This system has important advantages over incineration -when incineration is allowed-, such as: lack of ash generation, it can be located in the waste production plant -so no waste transport is needed-, and no dioxins are generated.
The advantages of this system are both environmental and economical. The environmental benefit lies in the high minimization of the volume of a nuclear residue that practically does not have radioactive activity. On the other hand, the economic value is related to the reduction of the volume of a waste that has to be managed following the nuclear procedures for low-level waste (LLW) storage.

© NUCLEANTECH 2021, All Right Reserved





