CONTENTS
These are the different sections in the presentation of our isotope adsorption solution. Visit any section directly by clicking the link.
1
ISOTOPE PRODUCTION IN PWR REACTORS
Most nuclear reactors operating across Europe are PWR reactors. They use enriched uranium fuel and are first cooled by a primary system transporting high-pressure water and temperature.
This extracts heat from the core generated by uranium fission and transfers it to a secondary system tasked with producing an electric current using a generator. The primary system does not interact with the exterior at any time, so the coolant circulating inside it is enriched over its running time by certain isotopic species due to the effect of several causes:
- The corrosion of the steel.
- The neutron activation of the corrosion products.
- Isotopes derived from the fuel rods.
In the first group we have Co60, Mn54, Co58 , among others, and in the second group we have Cs137 as a guiding element. The primary chemistry is very varied and complex.
Obviously all effluent involves a tritium solution from the neutron activation of the hydrogen and the use of lithium as a moderator element as fundamental causes.
2
ADSORBENTS: FUNCTION AND EFFECTIVENESS AGAINST RADIOACTIVE EFFLUENTS
In contrast to traditional resin treatment, Nucleantech supports the use of inorganic solid adsorbents for the treatment of radioactive effluents. These adsorbents present three high value-added properties for the treatment of radioactive effluents.
- The first property of these adsorbents is their selectivity with regards the isotope. We leverage a dedicated adsorbent for a particular isotope. Indeed, we can use mixtures of adsorbents to treat complex radioactive effluents (Co60, Co58, Cs137, Sb125, Mn54).
- The second property is their high efficacy in highly saline radioactive effluents. Most operational effluents (discharges, purges, ion exchange treatments, refuelling) normally contain a high level of salts: sodium chloride, borate, sulphates, etc. The saline ions compete with the resin exchange points and render them less effective.
One very specific case is Sb125. Treating this isotope requires a high resin level, since the salinity of the effluent competes with the antimony, which ends up flowing from the resin. All this leads to a high level of waste. The use of an adsorbent can reduce the waste volume, as antimony adsorption is not dependent on the salinity of the effluent. - A further fundamental feature of these adsorbents is their non-solubility in water, meaning they adsorb the isotope in question, and this precipitates with the adsorbent. This enables it to be filtered out of the solution.
3
SEPARATION OF ISOTOPES IN RADIOACTIVE EFFLUENTS BY MEANS OF ADSORBENTS
NUCLEANTECH has tested all these matters in a PWR nuclear plant with operational effluents from treatments, refuelling and conventional operations.
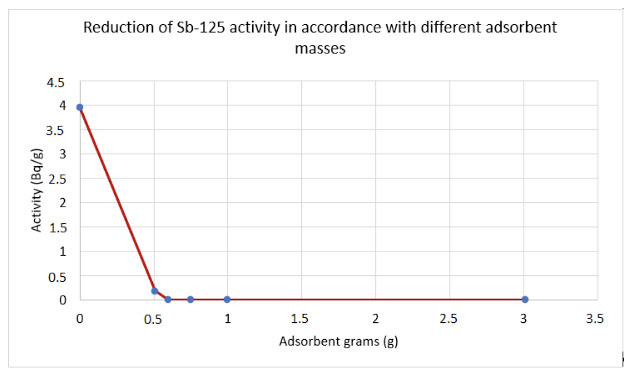
a.- Treatment of an effluent with Sb125 (4Bq/g); Co60 (0.67Bq/g); Cl-( (107mg/l); B(1200mg/l)
The presence of the antimony impedes resin treatment. Sb125 is a product of Sn124 activation present in zircaloy alloys.
In a pH range between 6 and 9, corresponding to the pH of the water stored in the tanks and in accordance with the Pourbaix diagram, the antimony is found in the form [Sb(OH)6]–.
Resin treatment is impeded by the presence of chlorides, sulphates and borates. Operational experience points to eluded activity 5 to 8 times greater than activity at bed entrance. The result is the early replacement of the resin mixture and a larger volume of waste.
El resultado es la sustitución temprana de la resina mixta y un mayor volumen de residuo.
Using the R3 adsorbent, specifically with a concentration of 6 g/l, and performing activity measurements by means of gamma spectrometry in the plant laboratory, Sb125 activity was almost entirely eliminated.
The results are shown on the chart below:
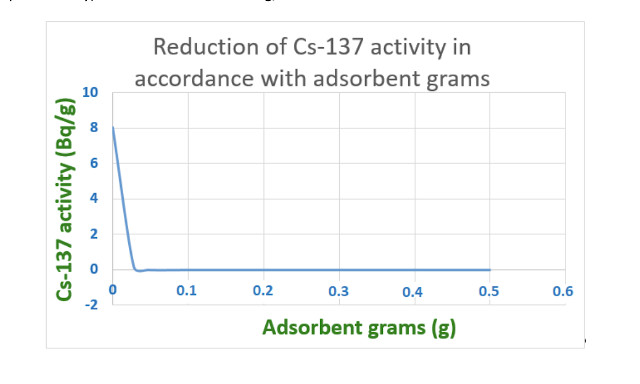
b.-Treatment of an effluent with Cs137, with an activity of 8 Bq/g
The use of a known adsorbent like R6 adsorbed 100 % of the isotope (no presence of the isotope was found by gamma spectrometry) in concentrations of 0.28 g/l.
4
TYPES OF ADSORBENTS
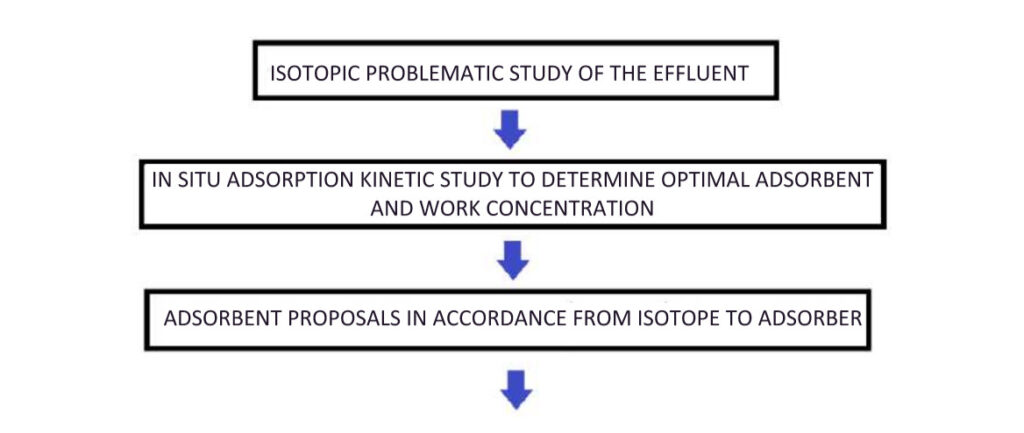
The choice of correct adsorbent to separate each type of isotope is made by following the procedure below:
Nucleantech has worked with different types of adsorbents, depending on the specific operational issue.
Effluents with Sb125, effluents with an isotopic mixture of Co60, Co58, operational effluents with a high content of Cs137.
It has also performed in-lab adsorption kinetics with Sr90, U238 and Am241.

The range of adsorbents is focused on 5 types:
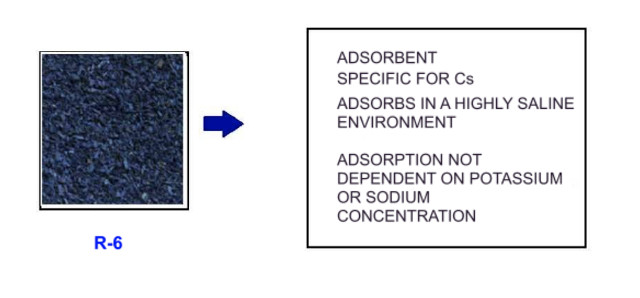
1.-R1 and R6 adsorbents. Adsorbents used for the treatment of Cs137. Adsorbents based on the hexacyanoferrate structure; КхFey[Fe(CN)6], where x = 0.2-0.8; y = 1.2-1.8.

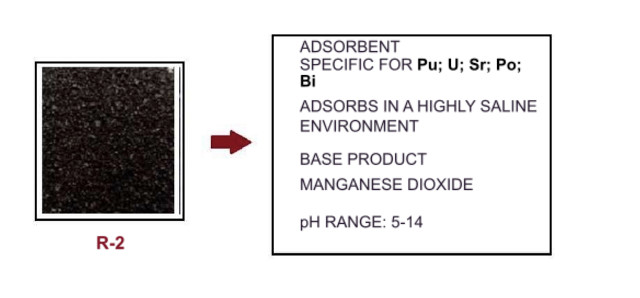
2.-R2 adsorbent. Based on MnO2 adsorption using a silicate base transporter. Used in the adsorption of Strontium, Uranium, Plutonium, Bismuth, Polonium.
3.-R3 adsorbent. KCuNi[Fe[CN]6]. Treatment specifically for Cs137.

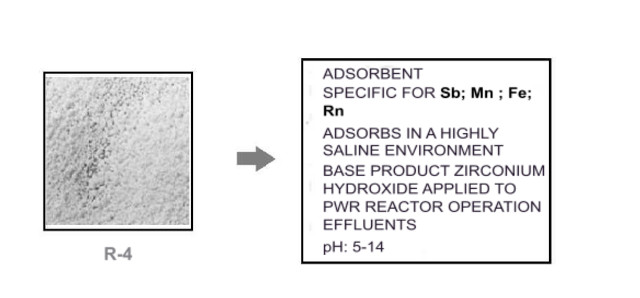
4.-R4 adsorbent. Based on the adsorption capacity of zirconium hydroxide. Used for alpha-emitting daughter isotopes.
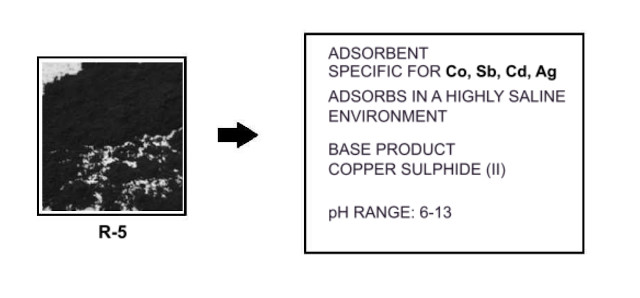
5.- R5 adsorbent. Based on fastening copper sulphides to silicate structures. Used for treating Ag110; Sb125; Co60; Co58.
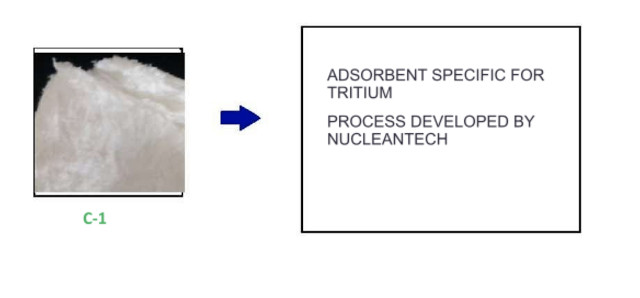
6.- C-1 adsorbent. Adsorbent developed exclusively for tritium separation. More information on our tritium separation solution
7.- Other adsorbents
5
RADIOACTIVE EFFLUENT TREATMENT USING TRADITIONAL PROCESSES
Regulatory systems are now starting to leverage resins (cationic, anionic and mixtures) as a core resource in activated wastewater treatment. Effluent is mainly cleaned from the primary system using resins. There are advantages and drawbacks to the use of these elements, as with all technology tools.
The main benefit is that the above mentioned isotopes are fastened.
The two main drawbacks are:
- Low efficacy in high salinity conditions to retain the isotopes (a much larger resin volume must be used).
- Increase of volume by coming into contact with the effluent (typical property of resins, in some cases 20 %).
These two drawbacks entail a higher waste volume (200 litre packages) and a cost increase we must all assume. This is particularly important given the challenge of nuclear dismantling that will start to take place in around the next 10 years.
Two fundamental questions will be:
- What volume of waste are we going to generate?
- What will it cost?
These two questions are obviously subject to the priority matter of safety. Maximum safety and minimum risk must be guaranteed at all times.






